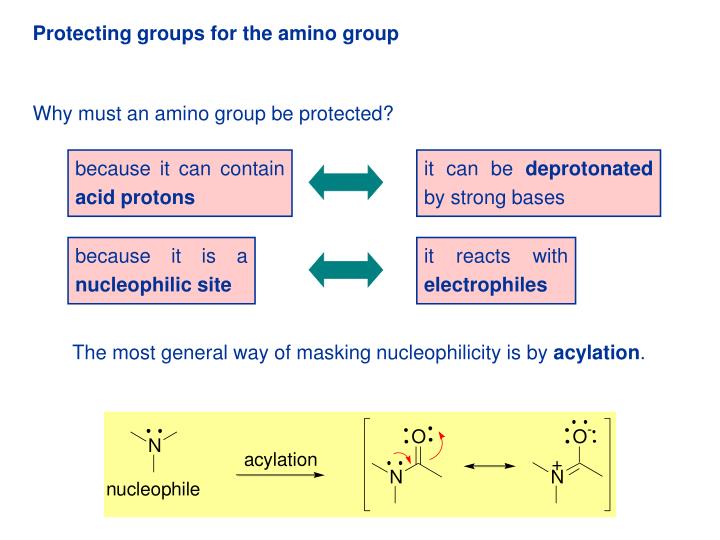
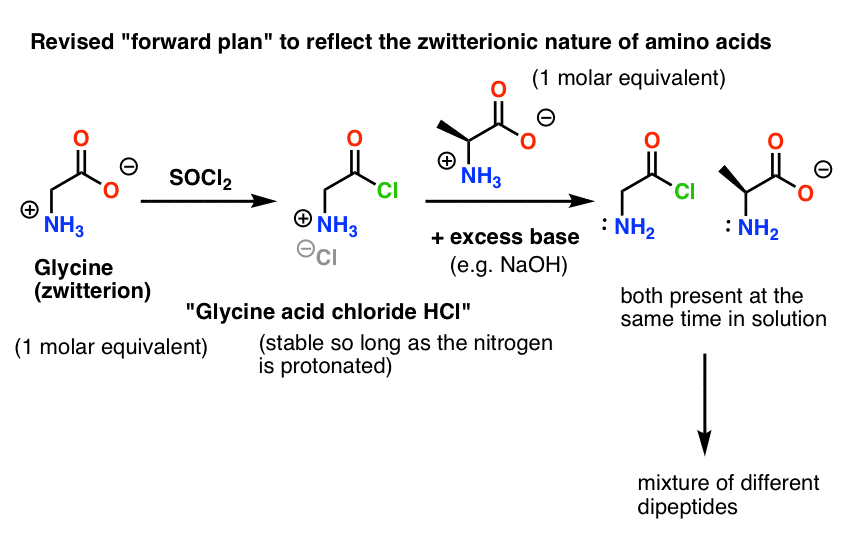
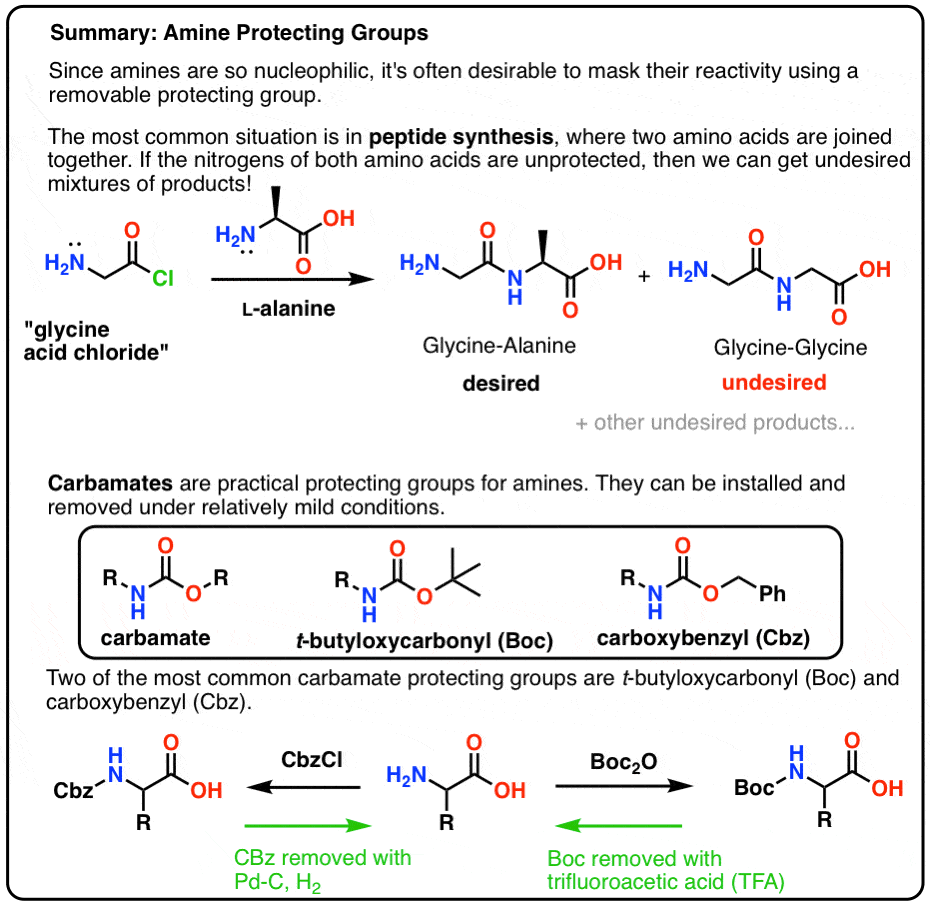
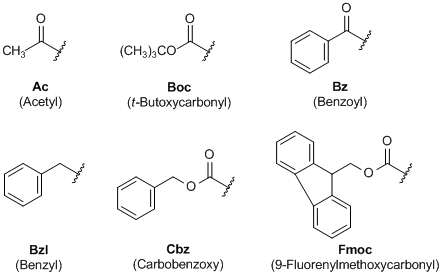
Unlocking the Potential of Phenacyl Protecting Groups: CO2-Based Formation and Photocatalytic Release of Caged Amines | The Journal of Organic Chemistry

Prevention of aspartimide formation during peptide synthesis using cyanosulfurylides as carboxylic acid-protecting groups | Nature Communications